Multiwavelength Telescope Astronomy in Studying Space
by SpaceHike.com
More articles in TelescopesHow do infrared pictures of people and X-rays of hands related to astronomy? Simple. Infrared cameras and X-ray film can give us new views of commonplace things on Earth, but similar techniques can be used to give us new information about things in space! Many things in space, like the Sun, the stars, our Galaxy, and other exotic objects like pulsars and black holes, emit light at many different energies.
Light has different colors because it has different energies. This is true whether we are talking about red and blue visible light, or infrared (IR) and X-ray light. Of all the colors in the visible spectrum, red light is the least energetic and blue is the most. Beyond the red end of the visible part of the spectrum lies infrared and radio light, both of which have lower energy than visible light. Above the blue end of the visible spectrum lies the higher energies of ultraviolet light, X-rays, and finally, gamma-rays.
How Do Telescopes Observe Different Energies of Light?
Optical telescopes are built to detect visible light waves and then to reflect the light using mirrors, or refract (bend) it with lenses, focusing it into an image. Because radio light is low energy, it tends to behave more like a wave, so most radio telescopes are dishes made of conducting metal that reflects radio waves to a focus, just as an optical mirror reflects visible light. Some infrared measurements can be made from the ground, but the majority of them are made from satellites to cut down on atmospheric absorption of infrared photons. X-rays and gamma-rays can only be collected from space because our atmosphere absorbs them.
When we compare measurements of the same region of the sky taken in different energy ranges, it becomes obvious how important multiwavelength observations are. Compare the below images, all of the entire sky, centered on our Galaxy. All are dominated by emission from our Galaxy, some from the Galactic center region (as in the X-ray image), others from the disk of our Galaxy (as in the infrared image).
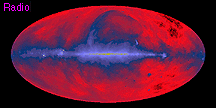 our galaxy's radio emissions
our galaxy's radio emissions our galaxy's infrared emissions
our galaxy's infrared emissions our galaxy's visible light emissions
our galaxy's visible light emissions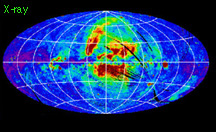 our galaxy's xray emissions
our galaxy's xray emissions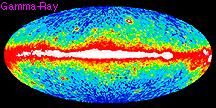 our galaxy's gamma ray emissions
our galaxy's gamma ray emissionsEnergy? Frequency? Wavelength?
Because of light's unique properties, it can be described in terms of either its energy, its wavelength, or its frequency. X-rays and gamma-rays are usually described in terms of energy, optical and infrared light in terms of wavelength, and radio in terms of frequency. This is a scientific convention that allows the use of the units that are the most convenient for describing whatever energy of light you are looking at. For example, it would be inconvenient to describe both low energy radio waves and high energy gamma-rays with the same units because the difference in their energies is so great (4 x 10-10 eV for radio as opposed to 4 x 109 eV for gamma-rays).
Wavelength is distance between two peaks of a wave, usually measured in terms of the base unit, the meter (m). The frequency of light is the number of cycles per second, or Hertz (Hz). Energy in astronomy is often measured in electron Volts, or eV. Wavelength and frequency are all related by the speed of light (which is a constant). Energy is also mathematically related to wavelength and frequency by a constant that we call Planck's constant (h) because Planck was the one to discover this relationship between them. This relationship says that energy (E) divided by frequency (the Greek letter nu) is a constant, and since we know that frequency is equal to the speed of light (c) divided by wavelength (the Greek letter lambda), we have the relationship between energy and wavelength.
In addition, light has what we call particle-wave duality, meaning that light can act like both a particle and a wave. The amount of energy a photon has makes it sometimes behave more like a wave and sometimes more like a particle. We call particles of light photons. Low energy photons (such as radio) tend to behave more like waves, while higher energy photons (such as X-rays) behave more like particles. This is an important difference because it affects the way we build instruments to measure light.